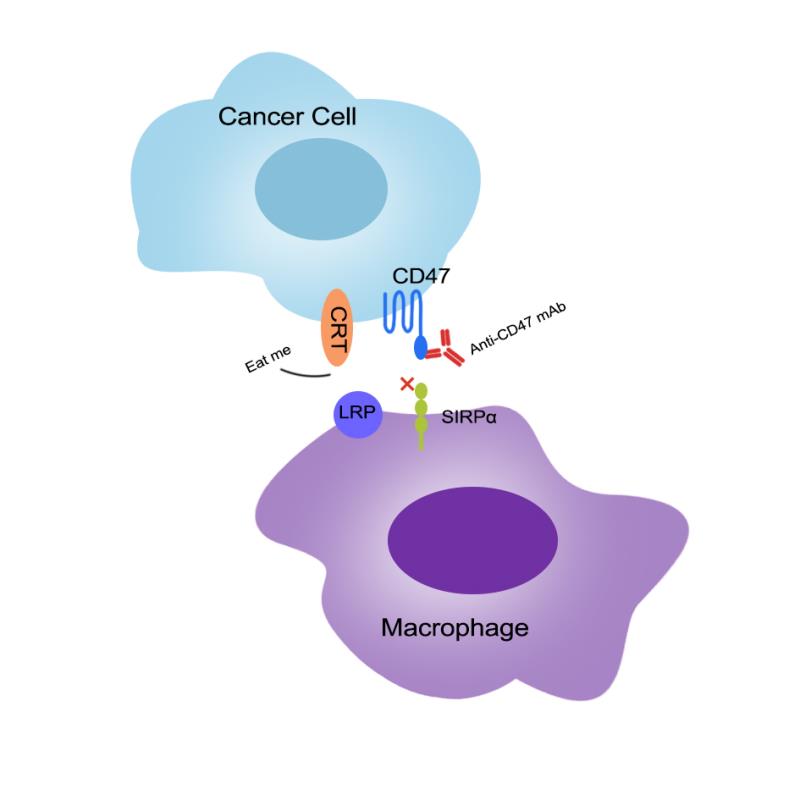
SIRPα, a transmembrane protein expressed on macrophages and bone marrow cells, binds to its ligand CD47 to produce a “don’t eat me” signal that prevents macrophages from phagocytizing healthy cells. Cancer cells are smart enough to utilize this mechanism to escape phagocytosis of macrophages. To specifically block this pathway will activate the tumor killing activity of macrophages.
(Source: DIMA BIOTECH)
At present, CD47 is relatively more favored target than its binding partner, the signal regulatory protein SIRPα. CD47 belongs to the immunoglobulin super family and is widely expressed in a variety of tumor and normal cells. Signal regulatory protein α (SIRPα) is a regulatory membrane glycoprotein from SIRP family expressed mainly by myeloid cells. Tumor cells can utilize the CD47-SIRPα signaling pathway to escape immune surveillance of macrophages .Therefore, blocking the binding of CD47 to SIRPα by anti-CD47 or anti-SIRPα therapeutic antibodies can eliminate the “don’t eat me” signal and activate the macrophage for killing tumor cells. Unfortunately, CD47 is also highly expressed on the surface of red blood cell membrane. Drugs targeting CD47 can kill tumor cells, but it also can cause anemia by injuring red blood cells. In addition, the widespread expression of CD47 will also create an “antigen sink” to make therapeutic antibody less effective. Right now, these are the two major challenges that anti-CD47 therapeutic antibodies have to face.
On the other hand, SIRPα exist only on the surface of macrophages and dendritic cells. Inhibition of SIRPα will not cause red blood cell anemia.
(Source: References[1])
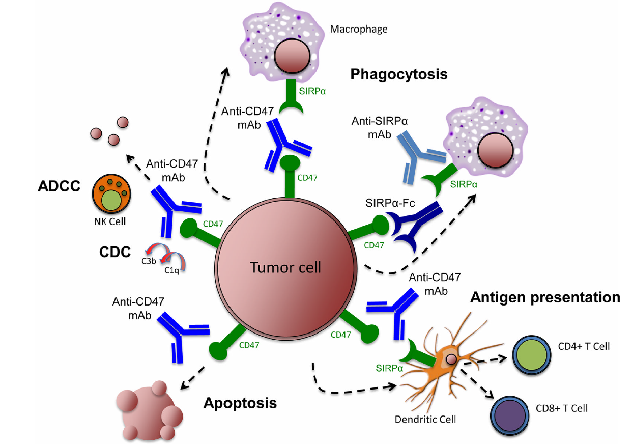
“Anti-SIRPα antibody immunotherapy enhances neutrophil and macrophage antitumor activity”, published in 2017 in PNAS, is a representative paper exploring the therapeutic option of anti-SIRPα antibodies. In this paper, the authors developed an anti-SIRPα mouse monoclonal antibody KWAR23, which can specifically block the binding of SIRPα to CD47. From their studies, they found that in vivo and in vitro, KWAR23 in combination with Rituximab, Obinuzumab, or other anti-CD20 antibodies, can significantly improve the phagocytosis to Burkitt’s lymphoma. In addition, KWAR23, when combined with EGFR antibodies, Cetuximab and Pantitumumab, can also enhance the phagocytosis of different colon cancer cell lines. Similarly, the bispecific monoclonal antibody, constructed from KWAR23 and an anti-CD70 therapeutic antibody, can also significantly inhibit tumor growth. However, it is quite surprising that anti-SIRPα antibody along can not induce the phagocytosis of tumor cells by macrophages.
Recently, a similar study was published in JCI insight, “Targeting macrophage checkpoint inhibitor SIRPα for anticancer therapy”. In this study, a new anti-SIRPα humanized monoclonal antibody 1H9 was developed by the group. This study demonstrated that 1H9 can specifically block the interaction between SIRPα and CD47, and also work synergistically with other therapeutic antibodies in vitro and in vivo.
From these studies, it is clear that SIRPα could be a good therapeutic drug target to develop combinatorial therapy or bispecific monoclonal antibody therapy.
In DIMA BIOTECH, we have successfully developed a good anti-SIRPα neutralizing antibody, by utilizing our rabbit monoclonal antibody development platform.
Anti-SIRPα (DM8) neutralizing antibody dataAntibody blocking data on THP-1 cells
Anti-SIRPα (DM8) can specifically block CD47-SIRPα interaction and the IC50 is 30.88ng/ml.
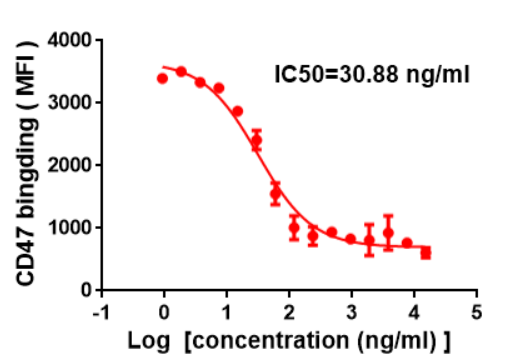
Figure 1. Competition assay demonstrating Anti-SIRPα DM8 can block CD47 protein binding to THP-1 cells. IC50=30.88ng/ml.
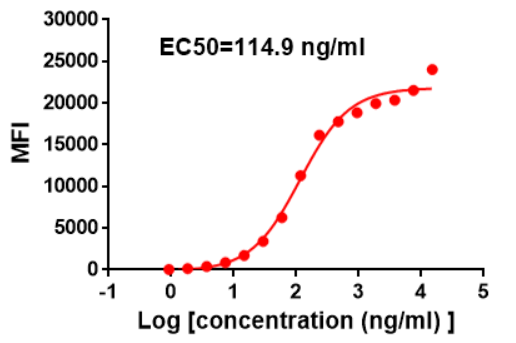
Figure 2. Flow cytometry data of serially titrated Anti-SIRPα (Clone: DM8). The Y-axis represents the mean fluorescence intensity (MFI) while the X-axis represents the concentration of IgG used.
Here, we would like to invite other interested companies forming partnership to co-develop therapeutic anti-SIRPα antibody.Right now, DM8 clone is listed on DIMA BIOTECH website and available for evaluation.
